Chemical Bonding – Overview
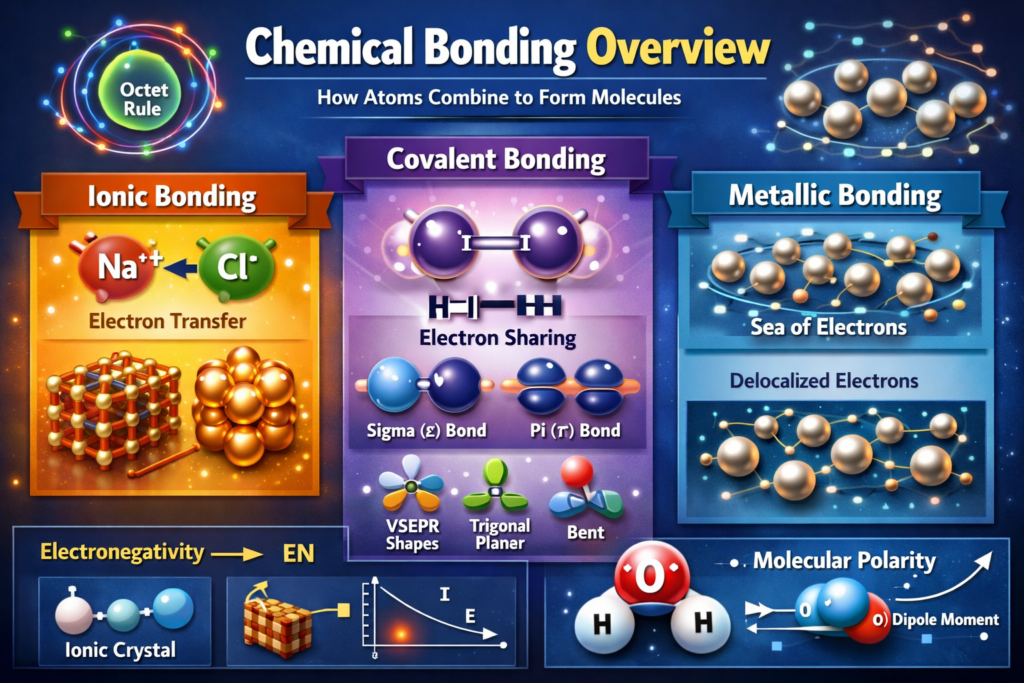
Chemical bonding explains how atoms combine to form molecules and compounds. The primary driving force behind bonding is the tendency of atoms to achieve a stable electronic configuration, often resembling the nearest noble gas (octet rule). The main types of chemical bonds include ionic bonds (transfer of electrons), covalent bonds (sharing of electrons), and metallic bonds (delocalized electrons). Concepts like electronegativity, ionization energy, and electron affinity help predict the nature of bonding between atoms.
In covalent bonding, atomic orbitals overlap to form molecular orbitals, leading to sigma (σ) and pi (π) bonds. Hybridization (sp, sp², sp³, etc.) explains molecular geometry and bond angles. Valence Shell Electron Pair Repulsion (VSEPR) theory predicts shapes of molecules based on electron pair repulsions. Bond parameters such as bond length, bond energy, and bond order are essential numerical aspects in chemical bonding.
Ionic bonding involves electrostatic attraction between oppositely charged ions, and its strength is quantified using lattice energy. Born-Haber cycles are used to calculate lattice energies indirectly. Additionally, dipole moment calculations help determine molecular polarity. These numerical aspects are critical in understanding stability, reactivity, and physical properties of compounds.
100 Practice Numerical Problems with Answers
Section A: Basic Concepts (1–20)
- Calculate the formal charge on O in H₂O.
Answer: 0 - Calculate bond order in O₂.
Answer: 2 - Bond order of N₂ molecule?
Answer: 3 - Number of bonds in CO₂?
Answer: 2 double bonds - Hybridization of carbon in CH₄?
Answer: sp³ - Bond angle in NH₃?
Answer: 107° - Dipole moment of CO₂?
Answer: 0 Debye - Hybridization of BeCl₂?
Answer: sp - Shape of BF₃?
Answer: Trigonal planar - Bond order of NO⁺?
Answer: 3 - Lone pairs in NH₃?
Answer: 1 - Bond angle in H₂O?
Answer: 104.5° - Shape of CH₄?
Answer: Tetrahedral - Hybridization in C₂H₂?
Answer: sp - Bond order of F₂?
Answer: 1 - Shape of SF₆?
Answer: Octahedral - Bond angle in CO₂?
Answer: 180° - Number of π bonds in C₂H₄?
Answer: 1 - Number of σ bonds in CH₄?
Answer: 4 - Bond order of O₂⁻?
Answer: 1.5
Section B: Lattice Energy & Ionic Bonding (21–40)
- Calculate lattice energy using Born-Haber data (given values).
Answer: Depends on data - Charge product of NaCl?
Answer: +1 × -1 = -1 - Which has higher lattice energy: MgO or NaCl?
Answer: MgO - Coulombic force proportional to?
Answer: q₁q₂/r² - Lattice energy trend: LiF vs LiCl?
Answer: LiF higher - Radius effect on lattice energy?
Answer: Inverse relation - Charge effect on lattice energy?
Answer: Direct relation - Lattice energy unit?
Answer: kJ/mol - Strongest ionic bond among NaCl, KCl, CsCl?
Answer: NaCl - Highest lattice energy: Al₂O₃ or NaCl?
Answer: Al₂O₃ - Born exponent relates to?
Answer: Repulsion - Madelung constant significance?
Answer: Crystal structure - Lattice energy increases with?
Answer: Charge - Smallest ion among Na⁺, K⁺, Li⁺?
Answer: Li⁺ - Highest melting point ionic compound?
Answer: MgO - Energy needed to separate ions?
Answer: Lattice energy - Ionic bond strength depends on?
Answer: Charge and distance - Larger anion → lattice energy?
Answer: Decreases - Ionic radius trend down group?
Answer: Increases - Electrostatic attraction formula constant?
Answer: Coulomb’s constant
Section C: Dipole Moment (41–60)
- Formula for dipole moment?
Answer: μ = q × d - Unit of dipole moment?
Answer: Debye - Dipole moment of symmetrical molecule?
Answer: Zero - NH₃ dipole moment?
Answer: Non-zero - HCl dipole moment direction?
Answer: Toward Cl - Polar molecule example?
Answer: H₂O - Nonpolar molecule example?
Answer: CO₂ - Dipole moment depends on?
Answer: Charge and distance - Larger electronegativity difference → μ?
Answer: Increases - μ of CH₄?
Answer: 0 - μ of SO₂?
Answer: Non-zero - μ of BF₃?
Answer: 0 - μ indicates?
Answer: Polarity - Unit conversion (C·m to Debye)?
Answer: 1 D = 3.336×10⁻³⁰ C·m - μ of linear symmetric molecule?
Answer: 0 - Vector sum concept applies?
Answer: Yes - Dipole moment increases with bond length?
Answer: Yes - Dipole moment zero means?
Answer: Nonpolar - CO dipole moment small due to?
Answer: Back bonding - μ highest in HF among HX?
Answer: Yes
Section D: Molecular Orbital Theory (61–80)
- Bond order formula?
Answer: (Nb – Na)/2 - Bond order of He₂?
Answer: 0 - Magnetic nature of O₂?
Answer: Paramagnetic - Magnetic nature of N₂?
Answer: Diamagnetic - Electrons in σ1s orbital of H₂?
Answer: 2 - Antibonding orbital symbol?
Answer: σ* - Bond order of O₂²⁻?
Answer: 1 - Stability proportional to?
Answer: Bond order - MO diagram includes?
Answer: Bonding & antibonding - Degenerate orbitals?
Answer: Equal energy - Bond order of NO?
Answer: 2.5 - Paramagnetism due to?
Answer: Unpaired electrons - Diamagnetism due to?
Answer: Paired electrons - Bond order of B₂?
Answer: 1 - π bonding from which orbitals?
Answer: p orbitals - σ bond stronger than π?
Answer: Yes - MO filling follows?
Answer: Aufbau principle - Hund’s rule applies?
Answer: Yes - Pauli principle applies?
Answer: Yes - Bond order fractional means?
Answer: Intermediate stability
Section E: Miscellaneous Numericals (81–100)
- Calculate formal charge on N in NH₄⁺.
Answer: +1 - Bond order relation to bond length?
Answer: Inverse - Hybridization in PCl₅?
Answer: sp³d - Shape of XeF₄?
Answer: Square planar - Lone pairs in XeF₄?
Answer: 2 - Bond angle in PCl₅?
Answer: 90° & 120° - Hybridization in SF₄?
Answer: sp³d - Shape of SF₄?
Answer: See-saw - Lone pairs in SF₄?
Answer: 1 - Hybridization in IF₇?
Answer: sp³d³ - Shape of IF₇?
Answer: Pentagonal bipyramidal - Bond order vs stability?
Answer: Direct - Strongest bond in N₂ or O₂?
Answer: N₂ - Number of electrons in valence shell?
Answer: Depends on element - Electron pair repulsion order?
Answer: LP-LP > LP-BP > BP-BP - Bond angle decreases with lone pairs?
Answer: Yes - Maximum covalency of sulfur?
Answer: 6 - Octet rule exceptions?
Answer: Yes - Polar bond vs polar molecule?
Answer: Different - Bond energy relation to bond order?
Answer: Direct