Molecular Geometry – Concept Overview
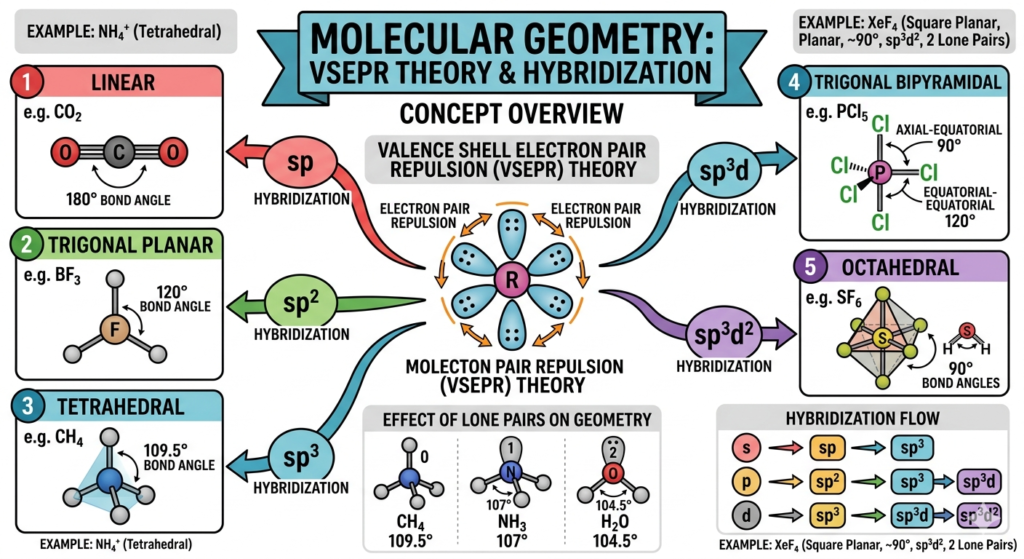
Molecular geometry refers to the three-dimensional arrangement of atoms in a molecule, which directly influences its physical and chemical properties such as polarity, reactivity, boiling point, and intermolecular interactions. The geometry is determined primarily by the number of bonding and lone electron pairs around the central atom. The most widely used model to predict molecular shapes is the Valence Shell Electron Pair Repulsion Theory, which states that electron pairs repel each other and arrange themselves as far apart as possible to minimize repulsion.
According to VSEPR theory, molecules adopt specific shapes such as linear, trigonal planar, tetrahedral, trigonal bipyramidal, and octahedral. For example, methane (CH₄) has a tetrahedral shape with bond angles of 109.5°, while carbon dioxide (CO₂) is linear with a bond angle of 180°. The presence of lone pairs alters bond angles due to stronger repulsion compared to bonding pairs, leading to shapes like bent (H₂O) and trigonal pyramidal (NH₃). Understanding these deviations is crucial for predicting real molecular behavior.
Hybridization of atomic orbitals also plays a key role in molecular geometry. It explains how atomic orbitals mix to form equivalent hybrid orbitals, such as sp, sp², sp³, sp³d, and sp³d², corresponding to different geometries. For instance, sp³ hybridization leads to tetrahedral geometry, while sp² leads to trigonal planar geometry. Combining VSEPR theory with hybridization concepts provides a strong foundation for solving numerical and conceptual problems in molecular geometry.
100 Practice Problems with Answers
Section A: Basic Geometry Identification (1–25)
- Determine the geometry of CO₂.
Answer: Linear - Shape of NH₃?
Answer: Trigonal pyramidal - Geometry of CH₄?
Answer: Tetrahedral - Shape of H₂O?
Answer: Bent - Geometry of BF₃?
Answer: Trigonal planar - Shape of PCl₅?
Answer: Trigonal bipyramidal - Geometry of SF₆?
Answer: Octahedral - Shape of SO₂?
Answer: Bent - Geometry of BeCl₂?
Answer: Linear - Shape of XeF₄?
Answer: Square planar - Geometry of NO₂⁻?
Answer: Bent - Shape of NH₄⁺?
Answer: Tetrahedral - Geometry of ClF₃?
Answer: T-shaped - Shape of ICl₂⁻?
Answer: Linear - Geometry of SF₄?
Answer: Seesaw - Shape of BrF₅?
Answer: Square pyramidal - Geometry of CO₃²⁻?
Answer: Trigonal planar - Shape of O₃?
Answer: Bent - Geometry of HCN?
Answer: Linear - Shape of CH₃Cl?
Answer: Tetrahedral - Geometry of PF₃?
Answer: Trigonal pyramidal - Shape of SO₃?
Answer: Trigonal planar - Geometry of NO₃⁻?
Answer: Trigonal planar - Shape of XeF₂?
Answer: Linear - Geometry of IF₅?
Answer: Square pyramidal
Section B: Bond Angle Calculations (26–50)
- Bond angle in CH₄?
Answer: 109.5° - Bond angle in NH₃?
Answer: 107° - Bond angle in H₂O?
Answer: 104.5° - Bond angle in BF₃?
Answer: 120° - Bond angle in CO₂?
Answer: 180° - Bond angle in PCl₅ (equatorial)?
Answer: 120° - Bond angle in PCl₅ (axial-equatorial)?
Answer: 90° - Bond angle in SF₆?
Answer: 90° - Bond angle in XeF₄?
Answer: 90° - Bond angle in SO₂?
Answer: ~119° - Bond angle in NO₂⁻?
Answer: ~115° - Bond angle in ClF₃?
Answer: ~87° - Bond angle in SF₄?
Answer: ~102° - Bond angle in BrF₅?
Answer: ~90° - Bond angle in O₃?
Answer: ~117° - Bond angle in CH₃Cl?
Answer: ~109.5° - Bond angle in NH₄⁺?
Answer: 109.5° - Bond angle in CO₃²⁻?
Answer: 120° - Bond angle in NO₃⁻?
Answer: 120° - Bond angle in HCN?
Answer: 180° - Bond angle in XeF₂?
Answer: 180° - Bond angle in IF₅?
Answer: ~90° - Bond angle in PF₃?
Answer: ~97° - Bond angle in SO₃?
Answer: 120° - Bond angle in ICl₂⁻?
Answer: 180°
Section C: Lone Pairs & Hybridization (51–75)
- Hybridization of CH₄?
Answer: sp³ - Hybridization of BF₃?
Answer: sp² - Hybridization of CO₂?
Answer: sp - Hybridization of NH₃?
Answer: sp³ - Hybridization of H₂O?
Answer: sp³ - Hybridization of PCl₅?
Answer: sp³d - Hybridization of SF₆?
Answer: sp³d² - Hybridization of SO₂?
Answer: sp² - Hybridization of NO₃⁻?
Answer: sp² - Hybridization of CO₃²⁻?
Answer: sp² - Lone pairs in NH₃?
Answer: 1 - Lone pairs in H₂O?
Answer: 2 - Lone pairs in XeF₄?
Answer: 2 - Lone pairs in ClF₃?
Answer: 2 - Lone pairs in SF₄?
Answer: 1 - Lone pairs in BrF₅?
Answer: 1 - Lone pairs in ICl₂⁻?
Answer: 3 - Lone pairs in XeF₂?
Answer: 3 - Lone pairs in NO₂⁻?
Answer: 1 - Lone pairs in SO₂?
Answer: 1 - Hybridization of XeF₄?
Answer: sp³d² - Hybridization of ClF₃?
Answer: sp³d - Hybridization of SF₄?
Answer: sp³d - Hybridization of BrF₅?
Answer: sp³d² - Hybridization of XeF₂?
Answer: sp³d
Section D: Advanced / Mixed Problems (76–100)
- Predict geometry of molecule with 2 bonding pairs, 0 lone pairs.
Answer: Linear - Predict geometry with 3 bonding pairs, 0 lone pairs.
Answer: Trigonal planar - Predict geometry with 4 bonding pairs, 0 lone pairs.
Answer: Tetrahedral - Predict geometry with 3 bonding pairs, 1 lone pair.
Answer: Trigonal pyramidal - Predict geometry with 2 bonding pairs, 2 lone pairs.
Answer: Bent - Predict geometry with 5 bonding pairs.
Answer: Trigonal bipyramidal - Predict geometry with 6 bonding pairs.
Answer: Octahedral - Geometry with 4 bonding pairs, 2 lone pairs.
Answer: Square planar - Geometry with 3 bonding pairs, 2 lone pairs.
Answer: T-shaped - Geometry with 4 bonding pairs, 1 lone pair.
Answer: Seesaw - Which has larger bond angle: NH₃ or H₂O?
Answer: NH₃ - Which is linear: CO₂ or SO₂?
Answer: CO₂ - Which has no lone pairs: BF₃ or NH₃?
Answer: BF₃ - Which is polar: CO₂ or H₂O?
Answer: H₂O - Which has trigonal planar geometry: SO₃ or NH₃?
Answer: SO₃ - Which has tetrahedral geometry: CH₄ or BF₃?
Answer: CH₄ - Which has highest bond angle: CH₄ or NH₃?
Answer: CH₄ - Which has more lone pairs: H₂O or NH₃?
Answer: H₂O - Which has octahedral geometry: SF₆ or PCl₅?
Answer: SF₆ - Which is bent: CO₂ or O₃?
Answer: O₃ - Which has sp hybridization: CO₂ or CH₄?
Answer: CO₂ - Which has sp² hybridization: BF₃ or NH₃?
Answer: BF₃ - Which has sp³ hybridization: CH₄ or CO₂?
Answer: CH₄ - Which is nonpolar: CO₂ or H₂O?
Answer: CO₂ - Which has square planar geometry: XeF₄ or SF₄?
Answer: XeF₄